Ex Vivo Model
Lymphomoids: A Tissue-based Ex Vivo Culture System for Lymphoma Therapy Screening
VitroGel® RGD High Concentration
RGD modified – tunable, xeno-free hydrogel – high concentration (3 mL kit)
VitroGel RGD High Concentration
VitroGel® RGD High Concentration is a tunable, xeno-free (animal origin-free) hydrogel system modified with cell adhesive peptide RGD to promote cell attachment and cell-matrix interactions during the 3D cell culture. VitroGel RGD High Concentration comes with VitroGel Dilution Solution to adjust the final hydrogel strength from 10 to 4000 Pa.
VitroGel High Concentration hydrogels are our xeno-free, tunable hydrogels for researchers wanting full control to manipulate the biophysical and biological properties of the cell culture environment. The tunability of the hydrogel gives the ability to create an optimized environment for cell growth. The hydrogel system has a neutral pH, transparent, permeable and compatible with different imaging systems. The solution transforms into a hydrogel matrix by simply mixing with the cell culture medium. No cross-linking agent is required. Cells cultured in this system can be easily harvested with our VitroGel® Cell Recovery Solution. The hydrogel can also be tuned to be injectable for in vivo studies.
From 3D cell culture, 2D cell coating to animal injection, VitroGel makes it possible to bridge the in vitro and in vivo studies with the same platform system.
Mix & Match – 3D Cell Culture Your WAY!
![]() Unique to VitroGel High Concentration hydrogels is the ability to tailor create a multi-functional hydrogel by blending different types of VitroGel. VitroGel® RGD High Concentration can be “mix & matched” with other VitroGel High Concentration hydrogels such as VitroGel® IKVAV, VitroGel® YIGSR, VitroGel® MMP, and VitroGel® COL to create a customized multi-functional hydrogel. Using this flexible and powerful hydrogel system, scientists customize their 3D culture micro-environment for different applications.
Unique to VitroGel High Concentration hydrogels is the ability to tailor create a multi-functional hydrogel by blending different types of VitroGel. VitroGel® RGD High Concentration can be “mix & matched” with other VitroGel High Concentration hydrogels such as VitroGel® IKVAV, VitroGel® YIGSR, VitroGel® MMP, and VitroGel® COL to create a customized multi-functional hydrogel. Using this flexible and powerful hydrogel system, scientists customize their 3D culture micro-environment for different applications.
Specifications
| Contents | VitroGel® RGD High Concentration, 3 mL VitroGel® Dilution Solution, 50 mL |
| Hydrogel Formulation | Xeno-free tunable hydrogel modified with RGD peptide. |
| Use | Good for adhesion cells or cells requiring stronger cell-matrix interactions. |
| Mix & Match | Can be blended with other versions of VitroGel concentrated hydrogels to create a custom multi-functional matrix. |
| Operation | Room temperature |
| Hydrogel Strength | 10 to 4,000 Pa of G’ depending on dilution ratio. Dilute with VitroGel Dilution Solution (TYPE 1 or TYPE 2) for different concentrations. |
| pH | Neutral |
| Color | Transparent |
| Cell Harvesting | VitroGel Organoid Recovery Solution 5-15 min cell recovery |
| Injectable | Injectable hydrogel |
| Storage | Store at 2-8°C. Ships at ambient temperature |
| Number of Uses | Dilution ratio: 1:2 = 225 uses at 50 µL per well 1:3 = 300 uses at 50 µL per well 1:5 = 450 uses at 50 µL per well |
3D Cell Culture Process in 20 Minutes
VitroGel High Concentration hydrogels are easy to use. There is no cross-linking agent required. Work confidently at room temperature.
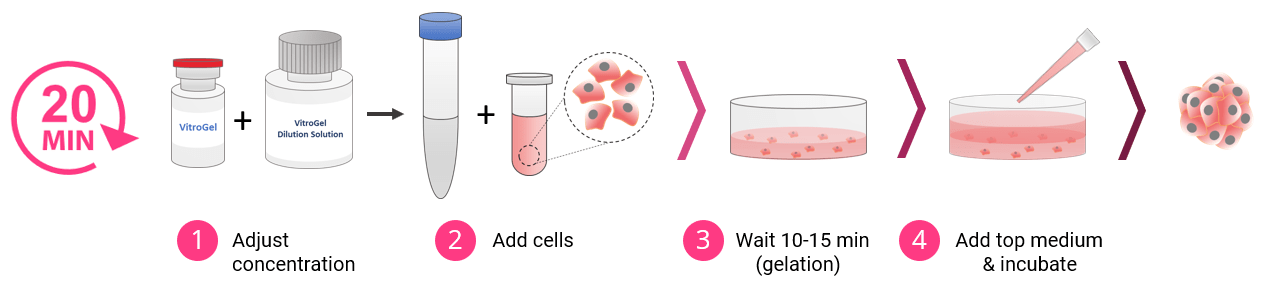
Tunable Hydrogel Strength
Simply diluting the hydrogel controls the gel strength.
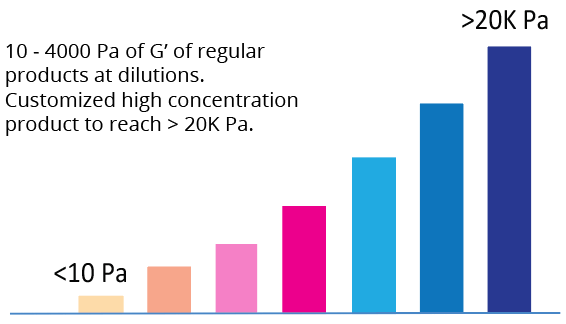
Protocols / Handbooks / Resources
Video Protocols & Demonstrations
Webinars
Research Highlights
Application Notes
Data and References
Cell Type Behavior Reference Table – VitroGel RGD
Multiple studies have made use of RGD hydrogel in different tissue and cell types. RGD is commonly used as an immobilized, adhesive ligand in 3D hydrogels that allows researcher to study many different cellular processes and behaviors in normal physiological and pathological contexts.
| Cell Type | Behavior |
|---|---|
| Goat bone marrow stromal cells | Promoted osteogenic differentiation |
| Rat bone marrow stromal cells | Promoted osteogenic differentiation |
| Rat osteoblasts | Increased cell attachment and spreading |
| Cell Type | Behavior |
|---|---|
| Biphasic synovial sarcoma SYO-1 | Cell proliferation and cell matrix interactions |
| Bone OSA 1777 | Cell proliferation and cell matrix interactions |
| Breast 4T1 | Cell proliferation, division, migration, and invasion |
| Breast AU-565 | Cell proliferation and cell matrix interactions |
| Breast Cancer MCF-7 | Cell proliferation, intercellular connections |
| Breast E0771 | Cell proliferation and cell matrix interactions |
| Breast MDA-MB-231 | Cell proliferation, division, migration, and invasion |
| Breast T47D | Cell proliferation, division, migration, and invasion |
| Colorectal adenocarcinoma DLD-1 cells | Cell proliferation and cell matrix interactions |
| Epithelial ovarian OV-MZ-6 | Promoted spheroid formation and proliferation |
| Epithelial ovarian SKOV-3 | Promoted spheroid formation and proliferation |
| Fuji Cells | Cell proliferation and cell matrix interactions |
| Glioblastoma SF 268 | Cell proliferation and cell matirx interaction |
| Glioblastoma SF 295 | Cell proliferation and cell matirx interaction |
| Glioblastoma SNB75 | Cell proliferation and cell matirx interaction |
| Glioblastoma U-251 MG | Cell proliferation and cell matirx interaction |
| Glioma U87-MG | Increased cell spreading and actin stress fiber assembly |
| Glioma U87-MG | Cell proliferation and cell matirx interaction |
| Glioma U373-MG | Increased cell adhesion duration and migration (on higher stiffness) |
| HEK 293 | Cell proliferation and cell matrix interactions |
| Huaman colon carcinoma HCT-8 | Cell proliferation and cell matirx interaction |
| Human colorectal carcinoma HCT 116 | cell proliferation, cell survival, and intercelluar networking |
| Human pancreatic cancer PANC-1 | cell proliferation and cellular interactions |
| Insulinoma ins-1 (Rat) | Cell proliferation and cell matrix interactions |
| Liver carcinoma HepG2 | Cell proliferation and cell matirx interaction |
| Melanoma Cells | Cell proliferation and cell matrix interactions |
| Ovarian carcinoma OVCAR-3 | Cell proliferation and invasion |
| Primary glioblastom U87 | cell proliferation and cellular interactions |
| Prostate adenocarcinoma LNCaP | Increased cell attachment |
| Prostate CRPC | Cell proliferatin and invasion |
| Prostate DU145 | Cell proliferation and invasion |
| Prostate PC3 | Cell proliferation and invasion |
| Cell Type | Behavior |
|---|---|
| Bovine chondrocytes | Increased cell viability and proliferation |
| Bovine chondrocytes | Promoted cell attachment, viability, and stress fiber formation |
| Human chondrocytes | Promoted cell viability and proliferation |
| Cell Type | Behavior |
|---|---|
| Fibroblast NIH3T3 | Promoted cell spreading |
| Fibroblasts NIH3T3 | Increased directional cell migration toward gradient |
| Human dermal fibroblasts | Promoted cell survival and spreading |
| Human dermal fibroblasts | Increased cell adhesion and proliferation |
| Human foreskin fibroblasts | Promoted cell spreading |
| Cell Type | Behavior |
|---|---|
| A549 cells | Cell proliferation and invasion |
| MCF-12A | Cell proliferation and invasion |
| Mouse ovarian follicle cells | Cell proliferation and invasion |
| Cell Type | Behavior |
|---|---|
| Beta TC3 Cells | Cell proliferation and cellular interactions |
| Cell Type | Behavior |
|---|---|
| Human embryonic kidney HEK293 | Promoted spheroid formation |
| Madin-Darby Canine Kidney | Promoted formation of structured epithelial cysts |
| Cell Type | Behavior |
|---|---|
| Human hepatocytes | Increased number of filopodia and synthesis of albumin |
| Mouse hepatocytes | Promoted cell viability |
| Cell Type | Behavior |
|---|---|
| Alveolar epithelial A549 | Inhibited cell detachment |
| Alveolar epithelial RLE-6TN | Increased cell attachment and mesenchymal differentiation |
| Cell Type | Behavior |
|---|---|
| Mouse skeletal myoblasts | Promoted cell attachment, proliferation, and myofibril formation |
| Myoblasts C2C12 | Promoted cell proliferation and differentiation |
| Cell Type | Behavior |
|---|---|
| Chick dorsal root ganglion cells | Increased neurite length, neurite outgrowth, and neurite number |
| In vivo lesioned rat cortex | Supported angiogenesis and inhibited glial scars |
| In vivo lesioned rat spinal cord | Supported angiogenesis and axon regeneration |
| Cell Type | Behavior |
|---|---|
| Human embryonic stem cells | Increased retinal pigmented epithelium and optic vesicle development |
| Human iPSC | Cell proliferation, and cell matrix interactions |
| Human mesenchymal stem cells | Increased cell viability |
| Mouse embryonic stem cells | Promoted endothelial cell differentiation |
| Mouse mesenchymal stem cells | Promoted cell spreading and migration |
| Rat mesenchymal stem cells | Increased cell adhesion and spreading |
| Rat mesenchymal stem cells | Promoted cell attachment and differentiation |
| Cell Type | Behavior |
|---|---|
| Human aortic smooth muscle cells | Promoted cell attachment |
| Human umbilical vein endothelial cells | Increased cell adhesion, proliferation, migration, and angiogenesis |
| Human umbilical vein endothelial cells | Increased cell adhesion and proliferation |
| Rat neonatal cardiac | Promoted cell attachment and tissue regeneration and prevented apoptosis |
| TISSUE/ORGAN TYPE | CELL TYPE | READY TO USE | HIGH CONCENTRATION | BEHAVIOR |
|---|---|---|---|---|
| Beta Cell | BL5 human beta cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D, VitroGel MMP | Enhance spheroids formation |
| Beta TC3 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cellular interactions | |
| Bone | Bone marrow stromal cells (rat) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Osteogensic differentiation , Cell attachment and osteoblast differentiation,Cell proliferation, cell viability, and cellular networking. |
| Osteoblasts (rat) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell attachment and spreading | |
| Bone marrow stromal cells (bovine) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell spreading and osteocalcin expression | |
| Breast | Mammary gland MCF10A | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitrolGel RGD, VitroGel COL, VitroGel MMP | Spheroid formation, MMP activity in response to TGF-B1 |
| Mammary epithelium (mouse) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell invasion and dissemination | |
| Cancer/Tumor | Human colorectal carcinoma HCT 116 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation, cell survival, and intercellular networking |
| Huaman colon carcinoma HCT-8 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Glioma U87-MG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP, VitroGel COL | Cell spreading and acting stress fiber assembly, cell proliferation, spreading, and migration, Cell migration dependent on mechancial force, Cell proliferation and cell matrix interaction | |
| Gliobastoma SF 268 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitrolGel RGD | Cell proliferation and cell matrix interaction | |
| Gliobastoma SF 295 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Glioblastoma SNB75 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Glioblastoma U-251 MG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Prostate PC3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL, VitroGel IKVAV, VitroGel RGD, VitroGel MMP | Cell proliferation, reduced MMP release, invasion, migration, and spheroid metabolic activity. | |
| Prostate LNCaP | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell attachment, proliferation, and prostate specific antigen release | |
| Prostate CRPC | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and invasion | |
| Prostate DU145 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and invasion | |
| Melanoma B16F10 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL, VitroGel YIGSR | Cell migration, invasion, MMP release, cell attachment and spreading | |
| Breast MDA-MB-231 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP, VitroGel 3D | Cell invasion, spreading, proliferation, division, migration, and cluster growth | |
| Fibrosarcoma HT1080 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell infiltration, attachment | |
| Breast T47D | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL, VitroGel 3D, VitroGel RGD, VitroGel MMP | Force dependent tubule formation, cell cluster growth, spheroid formation and proliferation | |
| Breast 4T1 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation | |
| Breast CTC | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D, VitroGel RGD | Cell proliferation | |
| Breast E0771 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation spheroid formation | |
| Brest AU-565 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation cell matrix interactions | |
| Epithelial ovarian OV-MZ-6 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Spheroid formation and proliferation | |
| Epithelial ovarian SKOV-3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Spheroid formation and proliferation | |
| Glioma U373-MG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell adhesion, invasion and migration | |
| Rhabdomyosarcoma (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel YIGSR | Cell attachment and spreading | |
| Melanoma SK-MEL-28 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Cell adhesion and proliferation | |
| Melanoma K-1735 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Cell invasion | |
| Melanoma A2058 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Collagenolytic activity | |
| Brainstem glioma DIPG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell proliferation and survival | |
| Hela Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D, VitroGel RGD, VitroGel MMP | Cell proliferation | |
| Colorectal adenocarcinoma DLD-1 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Giloma LRM55 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV, VitroGel MMP | Cell attachment | |
| Melanoma WM 239A | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell invasion | |
| Melanoma Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Insulinoma ins-1 (Rat) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Biphasic synovial sarcoma SYO-1 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation, cell matrix interation, and cell survival | |
| Fuji Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Chordoma Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Cell proliferation | |
| Bone OSA 1777 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Spheroid and cluster formation | |
| Glioma RuGli | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL | Integrin dependent cell adhesion | |
| Breast Cancer MCF-7 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP, VitroGel 3D | Cell proliferation, intercellular connections, morphological changes, MMP expression, and angiogenesis | |
| Liver carcinoma HepG2 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability, growth, drug resistance, proliferation, and cellular matrix interaction | |
| Human pancreatic cancer PANC-1 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell proliferation and cellular interactions | |
| Primary breast (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell invasion, migration, and dissemination | |
| Ovarian carcinoma OVCAR-3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP | Cell proliferation, cell matrix interactions | |
| Ovarian OVCA429 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP, VitroGel COL | MMP dependent cell invasion | |
| Human Osteosarcoma KHOS | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel 3D | Cell proliferation and spheroids formation | |
| Human Osteosarcoma U2OS | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel 3D | Cell proliferation and spheroids formation | |
| Human fibroblast-like synoviocytes (FLS) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Cell proliferation and inflammatory responses | |
| Human Liposarcoma 94T778 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Cell proliferation and spheroids formation | |
| Human diffuse large B-cell lymphoma (DLBLC) SUDHL-10 | VitroGel Hydrogel Matrix | Cell viability, growth, drug resistance, proliferation, and cellular matrix interaction | ||
| Priess human lymphoblastoid cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance spheroids and cluster formation and promote cell viability. | |
| Cartilage | Chondrocytes (bovine) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability and proliferation |
| Chondrocytes (human) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability and proliferation | |
| Connective Tissue | Dermal Fibroblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell viability and spreading |
| Fibroblasts NIH3T3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Directional cell migration toward gradient and cell spreading dependent on substrata rigidity | |
| Foreskin fibroblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel YIGSR, VitroGel MMP | Cell spreading, substrata degradation, and cell invasion | |
| Skin fibroblasts (skin) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV | Cell adhesion | |
| Epidermal keratinocytes | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability | |
| Epithelial Cells | Mouse ovarian follicle cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | 3D cell culture using ES-hydrogel can enhance vitro follicle culture by considering the permeability and stiffness of the gel. |
| Human Nthy-ori 3-1 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance spheroids and cluster formation and promote cell viability. | |
| A549 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Enhance cell proliferation and cell matrix interactions. | |
| MCF-12A | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Enhance cell proliferation and cell matrix interactions. | |
| Immortalized bronchial epithelial cells HBEC-KRAS | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel 3D | Cell proliferation | |
| Eye | Corneal endothelial B4G12 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit, VitroGel Angiogenesis Assay | VitroGel RGD, Vitrogel Angiogenesis Assay HC kit | Cell attachment and spreading |
| Retinal ganglion cells (xenopus) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Neurite outgrowth | |
| Immune Cells | CD8 + T cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance spheroids and cluster formation and promote cell viability. |
| Kidney | Human embryonic kidney HEK293 | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | 3D spheroids formation |
| Madin-Darby Canine Kidney | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP | Epithelial cysts formation | |
| Podocytes (human) | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Glomerular capillary formation |
|
| glomerular endothelial cells (human) | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit, VitroGel Angiogenesis Assay | VitroGel RGD, Vitrogel Angiogenesis Assay HC kit | Glomerular capillary formation |
|
| Liver | Hepatocytes (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Filopodia formation and synthesis of albumin and cell attachment |
| Hepatocytes (mouse, rat, swine) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell viability, spearding, Albumin secretion | |
| Lung | Alveolar basal epithelial A549 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell attachment |
| Alveolar epithelial RLE-6TN | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell attachment and mesenchymal differentiation | |
| Pulmonary fibroblasts LL2 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV | Cell adhesion | |
| HFL1 lung fibroblasts CCL153 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and spindle morphology | |
| Lung cancer associated fibroblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Substrata contractility | |
| Lung fibroblasts MCR-5 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | NGF-mediated substrata contraction | |
| Muscle | Myoblasts C2C12 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell proliferation, differentiation, attachment, myofibril formation, myotube formation, and integrin dependent cell adhesion |
| Skeletal myoblasts (mouse) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell attachment, proliferation, and myofibril formation | |
| Myoblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell adhesion, alignment along fiber, and myotube formation | |
| Myoblasts C25Cl48 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell proliferation, differentiation and myotube formation | |
| Neural | Dorsal root ganglion (chick) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Neurite formation and force dependent neurite outgrowth |
| Neural PC12 | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Neurite outgrowth | |
| Neural stem cell/ progenitor cell (rat) | VitroGel STEM, VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Cell viability, attachment, and differentiation | |
| Neural stem cell/ progenitor cell (human) | VitroGel STEM, VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV, VitroGel COL | Cell viability, attachment, and differentiation | |
| Schwann cells (rat) | VitroGel Hydrogel Matrix | VitroGel RGD | Cell attachment and migration | |
| Neural stem cell/ progenitor cell (mouse) | VitroGel STEM | VitroGel RGD, VitroGel IKVAV | Cell adhesion and differentiation | |
| Cortical astrocytes (rat) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Cell adhesion | |
| Spiral ganglion neurons (mouse) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Neurite outgrowth | |
| Motor neurons (human) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Force dependent neurite outgrowth | |
| Forebrain neurons (human) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Force dependent neurite outgrowth | |
| Cortical neurons (rat) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Neuronal viability and neurite outgrowth | |
| Dorsal root ganglion (rat) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Neurite outgrowth | |
| Red Blood Cells | Red Blood Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance Spheroids and cluster formation and promote cell viability |
| Pancreas | B-cells MIN6 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV | Reduced apoptosis and increased insulin release |
| Stem Cells | Mesenchymal stem cells (human) | VitroGel MSC | VitroGel RGD, VitroGel COL, VitroGel IKVAV, VitroGel MMP | Cell viability, proliferation, differentiation, neuronal differntiation, neurite outgrowth, attachment, spreading, viability, and osteoblast differentiation |
| Mesenchymal stem cells (mouse) | VitroGel MSC | VitroGel RGD, VitroGel MMP | Cell spreading and migration | |
| Mesenchymal stem cells (rat) | VitroGel MSC | VitroGel RGD | Cell adhesion and spreading | |
| Embryonic stem cells (mouse) | VitroGel STEM, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel YIGSR | Endothelial cell differentiation, neuronal differentiation, and neurite outgrowth | |
| Induced pluripotent stem cells (human) | VitroGel STEM, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel YIGSR, VitroGel IKVAV | Cell viability | |
| Human stem cells from apical papilla SCAP | VitroGel STEM | VitroGel RGD | Cell viability | |
| Hematopoietic Stem Cells | VitroGel STEM | Cell viability | ||
| Adipose derived stem cells (human) | VitroGel MSC | VitroGel RGD, VitroGel 3D, VitroGel IKVAV | Cell viability, cell attachment | |
| Vascular/cardiac | Umbilical vein endothelial cells | Vitrogel Angiogenesis Assay , VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | Vitrogel Angiogenesis Assay HC kit | Cell attachment, proliferation, migration, angiogenesis, gene expression changes, migratory cell infiltration, cell survival, and VEGF dependent migration |
| Neonatal cardiac (rat) | Vitrogel Angiogenesis Assay, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | Vitrogel Angiogenesis Assay HC kit | Cell attachment, tissue regeneration, and attachment similar to laminin | |
| Aortic smooth muscle cells | VitroGel Hydrogel Matrix | Vitrogel Angiogenesis Assay HC kit | Cell attachment | |
| Endothelial (human) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Cell differentiation | |
| Endotheliocytes | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Cell migration | |
| Microvascular endothelial cells (human) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Cell mobility | |
| Aortic endothelial cells (bovine) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Force dependent cell spreading | |
| Capillary endothelial cells (bovine) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Capillary like network formation |
Data
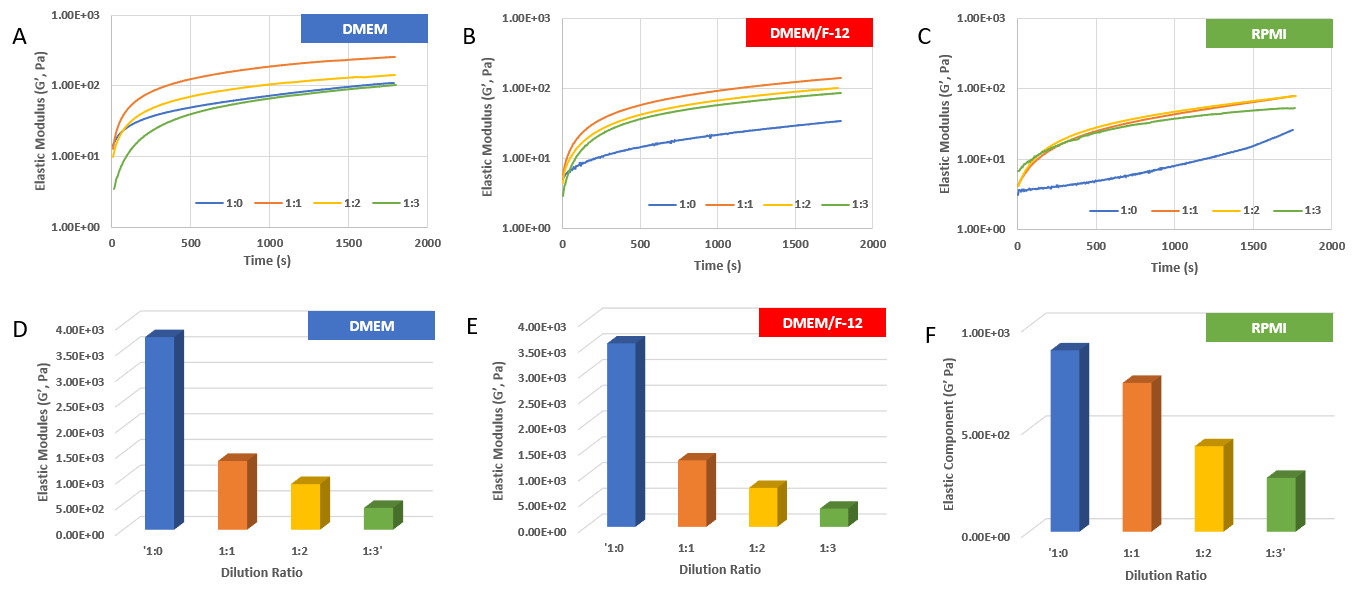
Figure 1. Rheological properties of VitroGel RGD with DMEM medium.
A) – C) The gel formation curve after mixing with DMEM (A), DMEM/F-12 (B), and RPMI (C) media. VitroGel RGD was diluted at 1:0,1:1, 1:2 and 1:3 (v/v) with VitroGel Dilution Solution (Type 1) and then mix with media at 4:1 (v/v) ratio; D) – F) The gel strength after 24 hrs incubation in DMEM (D), DMEM/F-12 (E), and RPMI (F) media. The hydrogel was prepared as method A and incubated at 37°C CO2 incubator for 24 hrs before the rheological test. (10 ~ 4000 Pa of G’ of regular products at dilutions. Customized high concentration product to reach over 20K Pa)

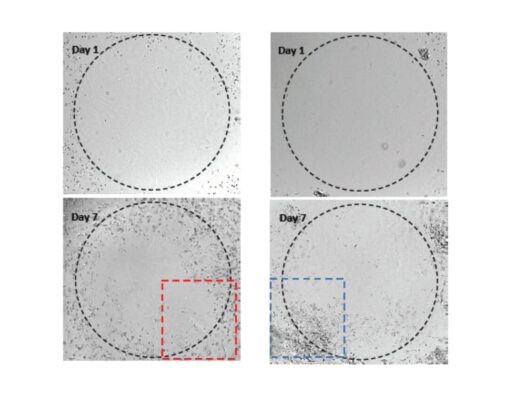
Figure 2. 3D culture of OP9 cells in VitroGel RGD.
Hydrogel was prepared at 1:3 dilution with VitroGel Dilution Solution (Type 1). The images were taken on days 2 and 7. VitroGel RGD shows support for OP9 cell proliferation and cell-cell communication. The stronger cell-matrix interactions help the cells to form the cell-networking structure.
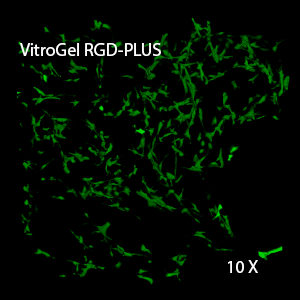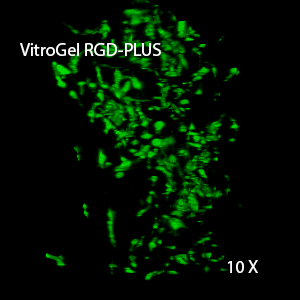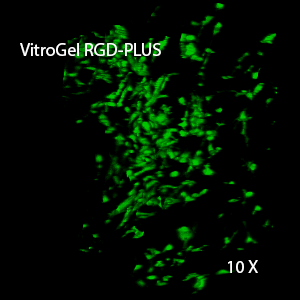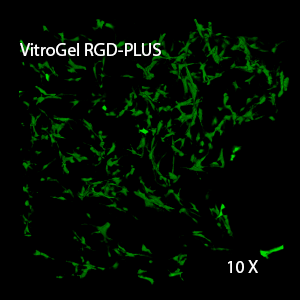
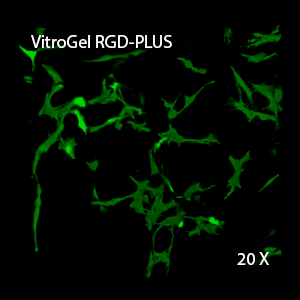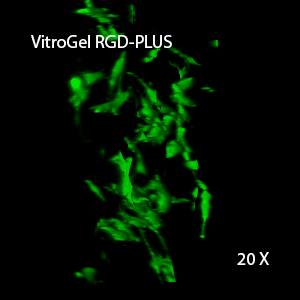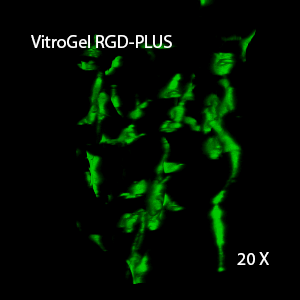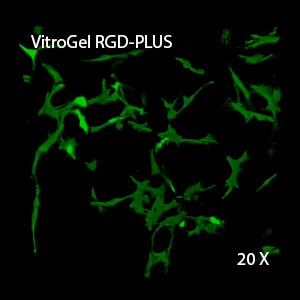
Figure 3. 3D view of OP9 cells growth in VitroGel RGD.
Cell networking structure formed in VitroGel RGD.

Figure 4. 3D culture of U87-MG cells in VitroGel RGD.
Cells can grow in 3D hydrogel at 1:1 and 1:3 dilution of VitroGel RGD. U87-MG cells shows cell networking structure and the cell morphology in VitroGel RGD indicating a cell-cell and cell-matrix interaction.
References/Publications
- Li, A., Huang, J., Chen, J., Wu, L., Zeng, H., Deng, Z., Liu, P., & Lin, J. (2025). Evolving Functional Hydrogel Strategies for Cartilage Engineering: From Fundamentals to Functional Regeneration. Burns & Trauma. https://doi.org/10.1093/burnst/tkaf041
- Wang, X., Stefanello, S. T., Shahin, V., & Qian, Y. (2025). From Mechanoelectric Conversion to Tissue Regeneration: Translational Progress in Piezoelectric Materials. Advanced Materials. https://doi.org/10.1002/adma.202417564
- Aimilia Zisiadi, Billooye, K., & Anckaert, E. (2025). A matrix-free 3D in vitro follicle culture system in mice exhibits enhanced oocyte meiotic and developmental competence compared to hydrogel encapsulation. Molecular Human Reproduction. https://doi.org/10.1093/molehr/gaaf029
- Sun, P., Qin, W., Xu, H., Yin, H., Yang, L., Zhang, X., Jin, X., Xu, Q., Wu, H., Xiaoling Kuai, Jia, L., Huang, J., & Wang, Y. (2025). SPTSSA facilitates gastric cancer progression with modulating PD-L1 in immunomicroenvironment through Wnt/β-catenin pathway. Cellular Oncology. https://doi.org/10.1007/s13402-025-01072-7
- Mokhtari, R. B., Sampath, D., Eversole, P., Ong, M., Bosykh, D. A., Gandhi T.K. Boopathy, Sivakumar, A., Wang, C., Kumar, R., Yeong, J., Karasik, E., Foster, B. A., Yu, H., Ling, X., Wu, W., Li, F., Ohler, Z. W., Brainson, C. F., Goodrich, D. W., & Hong, W. (2025). An Agrin–YAP/TAZ Rigidity Sensing Module Drives EGFR‐Addicted Lung Tumorigenesis. Advanced Science. https://doi.org/10.1002/advs.202413443
- Sun, P., Xu, H., Guo, C., Yang, L., Zhang, X., Lu, B., Chen, L., & Huang, J. (2025). TMEM115 as an Oncogenic and Immunological Biomarker in Hepatocellular Carcinoma. Liver International : Official Journal of the International Association for the Study of the Liver, 45(4), e70048. https://doi.org/10.1111/liv.70048
- Trucco, D., Gibney, R., Vannozzi, L., Lisignoli, G., Kelly, D. J., & Ricotti, L. (2025). Reinforcement of injectable hydrogels through melt electro-written structures: Influence of shape and pore size on the injection force. Journal of Materials Research and Technology, 36, 358–368. https://doi.org/10.1016/j.jmrt.2025.03.133
- Tschon, M., Codispoti, G., Cabras, P., Cafarelli, A., Trucco, D., Vannozzi, L., Manferdini, C., Carniato, M., Cassiolas, G., Martini, L., Fini, M., D’Atri, G., Jost, C., Fedutik, Y., Nessim, G. D., Dumont, E., Lisignoli, G., & Ricotti, L. (2025). In Vivo Efficacy of an Injectable Piezoelectric Nanocomposite Hydrogel and Low-Intensity Pulsed Ultrasound in Two Preclinical Models of Osteoarthritis.KeAi: Bioactive Materials https://doi.org/10.2139/ssrn.5128611
- Dindelegan, M. G., Blebea, C. M., Perde-Schrepler, M., Necula, V., Maniu, A. A., Pascalau, V., Popa, C., Susman, S., Gherman, L. M., & Buzoianu, A. D. (2024). Hydrogel Matrix Containing Microcarriers for Dexamethasone Delivery to Protect Against Cisplatin-Induced Hearing Loss. Cureus. https://doi.org/10.7759/cureus.71142
- Mhd Safwan Albougha, Hideki Sugii, Adachi, O., Mardini, B., Soeno, S., Hamano, S., Hasegawa, D., Yoshida, S., Tomohiro Itoyama, Obata, J., & Maeda, H. (2024). Exosomes from Human Periodontal Ligament Stem Cells Promote Differentiation of Osteoblast-like Cells and Bone Healing in Rat Calvarial Bone. Biomolecules, 14(11), 1455–1455. https://doi.org/10.3390/biom14111455
- Santamaria-Martínez, A., Epiney, J., Srivastava, D., Tavernari, D., Varrone, M., Milowich, D., Igor Letovanec, Krueger, T., Duran, R., Ciriello, G., Cairoli, A., & Oricchio, E. (2024). Development of patient-derived lymphomoids with preserved tumor architecture for lymphoma therapy screening. Nature Communications, 15(1). https://doi.org/10.1038/s41467-024-55098-w
- Haruna, N.-F., & Huang, J. (2020). Investigating The Dynamic Biophysical Properties Of A Tunable Hydrogel For 3D Cell Culture. HSOA Journal of Cytology and Tissue Biology. https://dx.doi.org/10.24966/CTB-9107/100030
- Gabusi, E., Lenzi, E., Manferdini, C., Dolzani, P., Columbaro, M., Saleh, Y., & Lisignoli, G. (2022). Autophagy Is a Crucial Path in Chondrogenesis of Adipose-Derived Mesenchymal Stromal Cells Laden in Hydrogel. Gels, 8(12), 766. https://doi.org/10.3390/gels8120766
- Hao, X., Zhang, S., Li, P., Huang, J., Yuan, Z., & Tan, J. (2022). Amniotic membrane extract-enriched hydrogel augments the therapeutic effect of menstrual blood-derived stromal cells in a rat model of intrauterine adhesion. Biomaterials Advances,142, 213165. https://doi.org/10.1016/j.bioadv.2022.213165
- Ding, J., et al. (2022). RGD-Hydrogel Improves the Therapeutic Effect of Bone Marrow-Derived Mesenchymal Stem Cells on Phosgene-Induced Acute Lung Injury in Rats Computational Intelligence and Neuroscience. https://www.hindawi.com/journals/cin/2022/2743878/
- Manferdini, C., et al. (2022). RGD-Functionalized Hydrogel Supports the Chondrogenic Commitment of Adipose Mesenchymal Stromal Cells Gels. https://www.mdpi.com/2310-2861/8/6/382
- Fen, et al.(2022) Optimization of Three-Dimensional Culture Conditions of HepG2 Cells with Response Surface Methodology Based on the VitroGel System. Biomedical and Environmental Sciences,(35,8), 688-698. https://www.frontiersin.org/articles/10.3389/fimmu.2022.914381/full
- Powell K. Adding depth to cell culture. Science, 356(6333), 96–98. https://doi.org/10.1126/science.356.6333.96
| High Concentration Kit Type | VitroGel RGD + Dilution Solution TYPE 1, VitroGel RGD + Dilution Solution TYPE 2 |
|---|