VitroGel® 3D High Concentration
tunable, xeno-free hydrogel, high concentration (3 mL kit)
VitroGel® 3D High Concentration
VitroGel® 3D High Concentration is a tunable, xeno-free (animal origin-free) hydrogel system that allows the maximum flexibility to manipulate the 3D cell culture environment for different needs. VitroGel® 3D High Concentration comes with VitroGel® Dilution Solution to adjust the final hydrogel strength from 10 to 4000 Pa. The hydrogel’s tunability gives researchers the ability to create an optimized environment for cell growth. The VitroGel® 3D High Concentration hydrogel matrix structure is good for cell spheroid formation, suspension cells, or cells requiring low cell-matrix interactions.
VitroGel® High Concentration hydrogels are our xeno-free, tunable hydrogels for researchers wanting full control to manipulate the biophysical and biological properties of the cell culture environment. The tunability of the hydrogel gives the ability to create an optimized environment for cell growth. The hydrogel system has a neutral pH, transparent, permeable, and compatible with different imaging systems. The solution transforms into a hydrogel matrix by simply mixing with the cell culture medium. No cross-linking agent is required. Cells cultured in this system can be easily harvested with our VitroGel® Cell Recovery Solution. The hydrogel can also be tuned to be injectable for in vivo studies.
From 3D cell culture, 2D cell coating to animal injection, VitroGel® makes it possible to bridge the in vitro and in vivo studies with the same platform system.
Specifications
| Contents | VitroGel® 3D High Concentration, 3 mL VitroGel® Dilution Solution, 50 mL |
| Hydrogel Formulation | Xeno-free tunable hydrogel, pure and unmodified. |
| Use | Good for cell spheroid formation, suspension cells or cells require low cell-matrix interactions |
| Mix & Match | Can be blended with other versions of VitroGel concentrated hydrogels to create a custom multi-functional matrix. |
| Operation | Room temperature |
| Hydrogel Strength | 10 to 4,000 Pa of G’ depending on dilution ratio. Dilute with VitroGel Dilution Solution (TYPE 1 or TYPE 2) for different concentrations. |
| pH | Neutral |
| Color | Transparent |
| Cell Harvesting | VitroGel Organoid Recovery Solution 5-15 min cell recovery |
| Injectable | Injectable hydrogel |
| Storage | Store at 2-8°C. Ships at ambient temperature |
| Number of Uses | Dilution ratio: 1:2 = 225 uses at 50 µL per well 1:3 = 300 uses at 50 µL per well 1:5 = 450 uses at 50 µL per well |
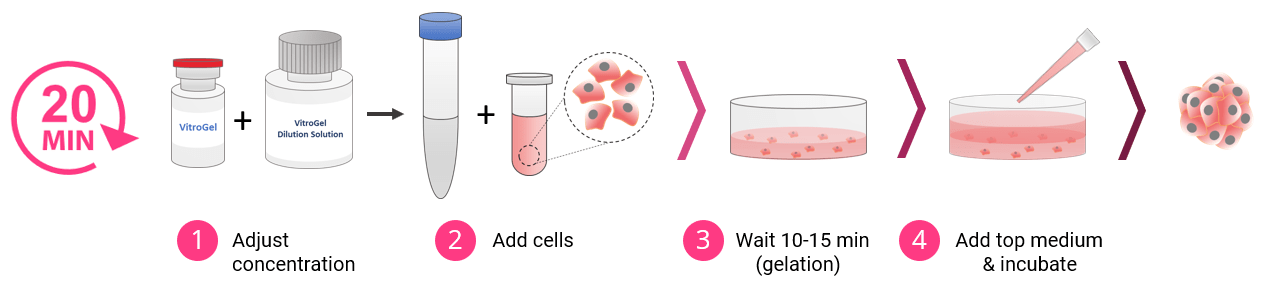
3D Cell Culture Process in 20 Minutes
VitroGel® High Concentration hydrogels are easy-to-use. There is no cross-linking agent required. Work confidently at room temperature.
Tunable Hydrogel Strength
Simply diluting the hydrogel controls the gel strength
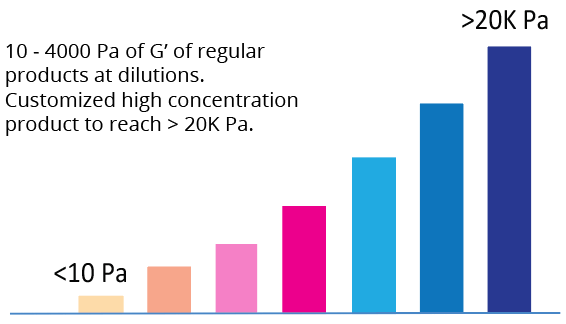
Protocols / Handbooks / Resources
Video Protocols & Demonstrations
Application Notes
Research Highlights
Data and References
Cell Type Behavior Reference Table – VitroGel 3D
Studies performed using VitroGel 3D in different tissue and cell types.
| Cell Type | Behavior |
|---|---|
| Brainstem glioma DIPG | Cell proliferation and survival |
| Breast CTC | Cell proliferation |
| Breast T47D | Spheroid formation and proliferation |
| Chordoma Cells | Cell proliferation |
| Hela Cells | Cell proliferation |
| Human osteosarcoma KHOS | Cell proliferation and spheroid formation |
| Human osteosarcoma U2OS | Cell proliferation and spheroid formation |
| Cell Type | Behavior |
|---|---|
| Human NTHY-ORI 3-1 Cells | Enhance spheroids and cluster formation and promote cell viability |
| Cell Type | Behavior |
|---|---|
| BL5 human beta cells | Enhance spheroids and cluster formation and promote cell viability |
| CD8 + T cells | Enhance spheroids and cluster formation and promote cell viability |
| Priess human lymphoblastoid cells | Enhance spheroids and cluster formation and promote cell viability |
| Cell Type | Behavior |
|---|---|
| Red Blood Cells | Enhance spheroids and cluster formation and promote cell viability |
| Cell Type | Behavior |
|---|---|
| Human stem cells from apical papilla SCAP | Enhance cell viability |
| TISSUE/ORGAN TYPE | CELL TYPE | READY TO USE | HIGH CONCENTRATION | BEHAVIOR |
|---|---|---|---|---|
| Beta Cell | BL5 human beta cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D, VitroGel MMP | Enhance spheroids formation |
| Beta TC3 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cellular interactions | |
| Bone | Bone marrow stromal cells (rat) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Osteogensic differentiation , Cell attachment and osteoblast differentiation,Cell proliferation, cell viability, and cellular networking. |
| Osteoblasts (rat) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell attachment and spreading | |
| Bone marrow stromal cells (bovine) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell spreading and osteocalcin expression | |
| Breast | Mammary gland MCF10A | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitrolGel RGD, VitroGel COL, VitroGel MMP | Spheroid formation, MMP activity in response to TGF-B1 |
| Mammary epithelium (mouse) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell invasion and dissemination | |
| Cancer/Tumor | Human colorectal carcinoma HCT 116 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation, cell survival, and intercellular networking |
| Huaman colon carcinoma HCT-8 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Glioma U87-MG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP, VitroGel COL | Cell spreading and acting stress fiber assembly, cell proliferation, spreading, and migration, Cell migration dependent on mechancial force, Cell proliferation and cell matrix interaction | |
| Gliobastoma SF 268 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitrolGel RGD | Cell proliferation and cell matrix interaction | |
| Gliobastoma SF 295 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Glioblastoma SNB75 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Glioblastoma U-251 MG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Prostate PC3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL, VitroGel IKVAV, VitroGel RGD, VitroGel MMP | Cell proliferation, reduced MMP release, invasion, migration, and spheroid metabolic activity. | |
| Prostate LNCaP | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell attachment, proliferation, and prostate specific antigen release | |
| Prostate CRPC | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and invasion | |
| Prostate DU145 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and invasion | |
| Melanoma B16F10 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL, VitroGel YIGSR | Cell migration, invasion, MMP release, cell attachment and spreading | |
| Breast MDA-MB-231 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP, VitroGel 3D | Cell invasion, spreading, proliferation, division, migration, and cluster growth | |
| Fibrosarcoma HT1080 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell infiltration, attachment | |
| Breast T47D | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL, VitroGel 3D, VitroGel RGD, VitroGel MMP | Force dependent tubule formation, cell cluster growth, spheroid formation and proliferation | |
| Breast 4T1 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation | |
| Breast CTC | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D, VitroGel RGD | Cell proliferation | |
| Breast E0771 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation spheroid formation | |
| Brest AU-565 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation cell matrix interactions | |
| Epithelial ovarian OV-MZ-6 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Spheroid formation and proliferation | |
| Epithelial ovarian SKOV-3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Spheroid formation and proliferation | |
| Glioma U373-MG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell adhesion, invasion and migration | |
| Rhabdomyosarcoma (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel YIGSR | Cell attachment and spreading | |
| Melanoma SK-MEL-28 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Cell adhesion and proliferation | |
| Melanoma K-1735 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Cell invasion | |
| Melanoma A2058 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Collagenolytic activity | |
| Brainstem glioma DIPG | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell proliferation and survival | |
| Hela Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D, VitroGel RGD, VitroGel MMP | Cell proliferation | |
| Colorectal adenocarcinoma DLD-1 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Giloma LRM55 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV, VitroGel MMP | Cell attachment | |
| Melanoma WM 239A | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell invasion | |
| Melanoma Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Insulinoma ins-1 (Rat) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Biphasic synovial sarcoma SYO-1 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation, cell matrix interation, and cell survival | |
| Fuji Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and cell matrix interaction | |
| Chordoma Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Cell proliferation | |
| Bone OSA 1777 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Spheroid and cluster formation | |
| Glioma RuGli | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel COL | Integrin dependent cell adhesion | |
| Breast Cancer MCF-7 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP, VitroGel 3D | Cell proliferation, intercellular connections, morphological changes, MMP expression, and angiogenesis | |
| Liver carcinoma HepG2 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability, growth, drug resistance, proliferation, and cellular matrix interaction | |
| Human pancreatic cancer PANC-1 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell proliferation and cellular interactions | |
| Primary breast (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell invasion, migration, and dissemination | |
| Ovarian carcinoma OVCAR-3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP | Cell proliferation, cell matrix interactions | |
| Ovarian OVCA429 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP, VitroGel COL | MMP dependent cell invasion | |
| Human Osteosarcoma KHOS | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel 3D | Cell proliferation and spheroids formation | |
| Human Osteosarcoma U2OS | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel 3D | Cell proliferation and spheroids formation | |
| Human fibroblast-like synoviocytes (FLS) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Cell proliferation and inflammatory responses | |
| Human Liposarcoma 94T778 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Cell proliferation and spheroids formation | |
| Human diffuse large B-cell lymphoma (DLBLC) SUDHL-10 | VitroGel Hydrogel Matrix | Cell viability, growth, drug resistance, proliferation, and cellular matrix interaction | ||
| Priess human lymphoblastoid cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance spheroids and cluster formation and promote cell viability. | |
| Cartilage | Chondrocytes (bovine) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability and proliferation |
| Chondrocytes (human) | VitroGel MSC, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability and proliferation | |
| Connective Tissue | Dermal Fibroblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell viability and spreading |
| Fibroblasts NIH3T3 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Directional cell migration toward gradient and cell spreading dependent on substrata rigidity | |
| Foreskin fibroblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel YIGSR, VitroGel MMP | Cell spreading, substrata degradation, and cell invasion | |
| Skin fibroblasts (skin) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV | Cell adhesion | |
| Epidermal keratinocytes | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell viability | |
| Epithelial Cells | Mouse ovarian follicle cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | 3D cell culture using ES-hydrogel can enhance vitro follicle culture by considering the permeability and stiffness of the gel. |
| Human Nthy-ori 3-1 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance spheroids and cluster formation and promote cell viability. | |
| A549 cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Enhance cell proliferation and cell matrix interactions. | |
| MCF-12A | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Enhance cell proliferation and cell matrix interactions. | |
| Immortalized bronchial epithelial cells HBEC-KRAS | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel 3D | Cell proliferation | |
| Eye | Corneal endothelial B4G12 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit, VitroGel Angiogenesis Assay | VitroGel RGD, Vitrogel Angiogenesis Assay HC kit | Cell attachment and spreading |
| Retinal ganglion cells (xenopus) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Neurite outgrowth | |
| Immune Cells | CD8 + T cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance spheroids and cluster formation and promote cell viability. |
| Kidney | Human embryonic kidney HEK293 | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | 3D spheroids formation |
| Madin-Darby Canine Kidney | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel MMP | Epithelial cysts formation | |
| Podocytes (human) | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Glomerular capillary formation |
|
| glomerular endothelial cells (human) | VitroGel Hydrogel Matrix, VitroGel HEK293, VitroGel ORGANOID Kit, VitroGel Angiogenesis Assay | VitroGel RGD, Vitrogel Angiogenesis Assay HC kit | Glomerular capillary formation |
|
| Liver | Hepatocytes (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Filopodia formation and synthesis of albumin and cell attachment |
| Hepatocytes (mouse, rat, swine) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel MMP | Cell viability, spearding, Albumin secretion | |
| Lung | Alveolar basal epithelial A549 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell attachment |
| Alveolar epithelial RLE-6TN | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell attachment and mesenchymal differentiation | |
| Pulmonary fibroblasts LL2 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV | Cell adhesion | |
| HFL1 lung fibroblasts CCL153 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell proliferation and spindle morphology | |
| Lung cancer associated fibroblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Substrata contractility | |
| Lung fibroblasts MCR-5 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | NGF-mediated substrata contraction | |
| Muscle | Myoblasts C2C12 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell proliferation, differentiation, attachment, myofibril formation, myotube formation, and integrin dependent cell adhesion |
| Skeletal myoblasts (mouse) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD | Cell attachment, proliferation, and myofibril formation | |
| Myoblasts (human) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell adhesion, alignment along fiber, and myotube formation | |
| Myoblasts C25Cl48 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Cell proliferation, differentiation and myotube formation | |
| Neural | Dorsal root ganglion (chick) | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL | Neurite formation and force dependent neurite outgrowth |
| Neural PC12 | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Neurite outgrowth | |
| Neural stem cell/ progenitor cell (rat) | VitroGel STEM, VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Cell viability, attachment, and differentiation | |
| Neural stem cell/ progenitor cell (human) | VitroGel STEM, VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV, VitroGel COL | Cell viability, attachment, and differentiation | |
| Schwann cells (rat) | VitroGel Hydrogel Matrix | VitroGel RGD | Cell attachment and migration | |
| Neural stem cell/ progenitor cell (mouse) | VitroGel STEM | VitroGel RGD, VitroGel IKVAV | Cell adhesion and differentiation | |
| Cortical astrocytes (rat) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Cell adhesion | |
| Spiral ganglion neurons (mouse) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel IKVAV | Neurite outgrowth | |
| Motor neurons (human) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Force dependent neurite outgrowth | |
| Forebrain neurons (human) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Force dependent neurite outgrowth | |
| Cortical neurons (rat) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Neuronal viability and neurite outgrowth | |
| Dorsal root ganglion (rat) | VitroGel Hydrogel Matrix | VitroGel RGD, VitroGel COL, VitroGel IKVAV | Neurite outgrowth | |
| Red Blood Cells | Red Blood Cells | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel 3D | Enhance Spheroids and cluster formation and promote cell viability |
| Pancreas | B-cells MIN6 | VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel IKVAV | Reduced apoptosis and increased insulin release |
| Stem Cells | Mesenchymal stem cells (human) | VitroGel MSC | VitroGel RGD, VitroGel COL, VitroGel IKVAV, VitroGel MMP | Cell viability, proliferation, differentiation, neuronal differntiation, neurite outgrowth, attachment, spreading, viability, and osteoblast differentiation |
| Mesenchymal stem cells (mouse) | VitroGel MSC | VitroGel RGD, VitroGel MMP | Cell spreading and migration | |
| Mesenchymal stem cells (rat) | VitroGel MSC | VitroGel RGD | Cell adhesion and spreading | |
| Embryonic stem cells (mouse) | VitroGel STEM, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel COL, VitroGel YIGSR | Endothelial cell differentiation, neuronal differentiation, and neurite outgrowth | |
| Induced pluripotent stem cells (human) | VitroGel STEM, VitroGel ORGANOID Kit | VitroGel RGD, VitroGel YIGSR, VitroGel IKVAV | Cell viability | |
| Human stem cells from apical papilla SCAP | VitroGel STEM | VitroGel RGD | Cell viability | |
| Hematopoietic Stem Cells | VitroGel STEM | Cell viability | ||
| Adipose derived stem cells (human) | VitroGel MSC | VitroGel RGD, VitroGel 3D, VitroGel IKVAV | Cell viability, cell attachment | |
| Vascular/cardiac | Umbilical vein endothelial cells | Vitrogel Angiogenesis Assay , VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | Vitrogel Angiogenesis Assay HC kit | Cell attachment, proliferation, migration, angiogenesis, gene expression changes, migratory cell infiltration, cell survival, and VEGF dependent migration |
| Neonatal cardiac (rat) | Vitrogel Angiogenesis Assay, VitroGel Hydrogel Matrix, VitroGel ORGANOID Kit | Vitrogel Angiogenesis Assay HC kit | Cell attachment, tissue regeneration, and attachment similar to laminin | |
| Aortic smooth muscle cells | VitroGel Hydrogel Matrix | Vitrogel Angiogenesis Assay HC kit | Cell attachment | |
| Endothelial (human) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Cell differentiation | |
| Endotheliocytes | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Cell migration | |
| Microvascular endothelial cells (human) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Cell mobility | |
| Aortic endothelial cells (bovine) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Force dependent cell spreading | |
| Capillary endothelial cells (bovine) | VitroGel Angiogenesis Assay | Vitrogel Angiogenesis Assay HC kit | Capillary like network formation |
Data
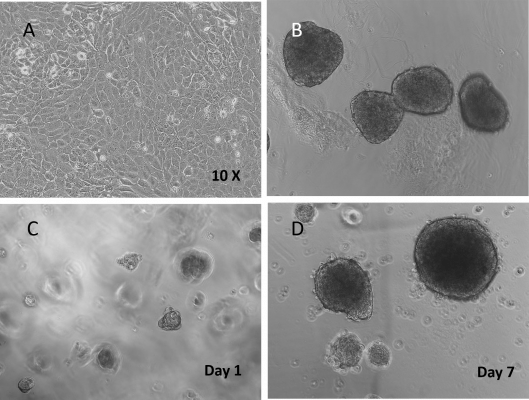
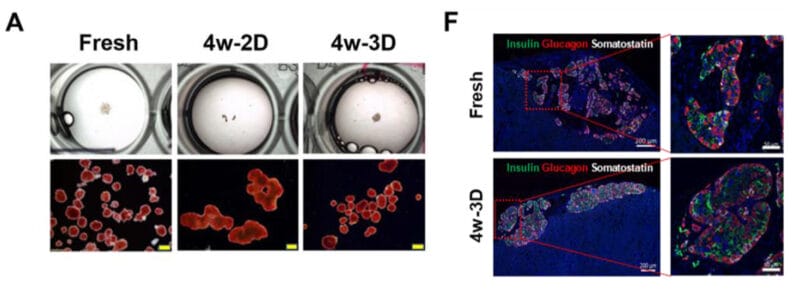
Figure 1. Beta Lox 5 (BL5) cells 3D culture in VitroGel 3D system.
A. BL5 cells culture on the surface of regular tissue culture treated well plate (control); B. Normal human islets grew in suspension culture (comparison); C. 3D culture of BL5 cells in VitroGel 3D at Day 1; D. 3D culture of BL5 cells in VitroGel 3D at Day 7. Under 3D culture of VitroGel 3D, BL5 cells form islet-like structures very similar to normal human islets. The hydrogel is prepared at 1:3 dilution. The images were taken at 10X magnification.
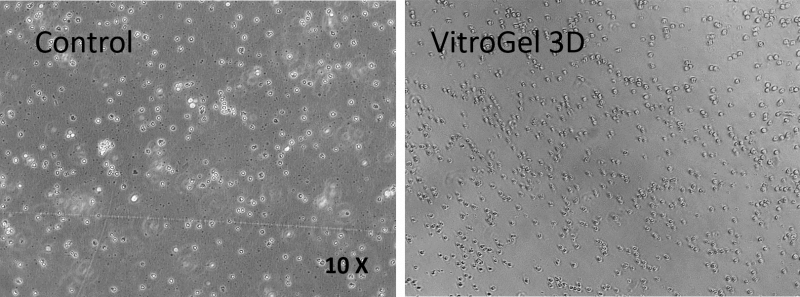
Figure 2. CD8+ T cells 3D culture in VitroGel 3D system.
CD8+ T cells culture grew in suspension culture (control); B. 3D culture of CD8+ T cells in VitroGel 3D at Day 7. CD8+ T cells are vibrant in 3D culture conditions of VitroGel 3D. The cells can easily move within the unmodified hydrogel matrix. The hydrogel is prepared at 1:3 dilution. The images were taken at 10X magnification.
2D Coating Applications
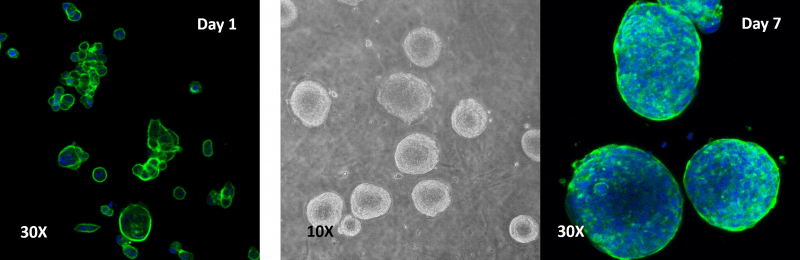
Figure 3. Human colon cancer cells (HCT 116) cells cultured on top of VitroGel 3D hydrogel.
A thick hydrogel coating plate has been prepared by mixing VitroGel 3D with PBS at 1:1 ratio. A 300 µL mixture has been added to a well of a 24-well plate and stabilization at room temperature for 20 minutes before adding cells on top of the hydrogel. Cell spheroids form on the top of the hydrogel. Cells seeded at 2.5-10×105 cells/mL.
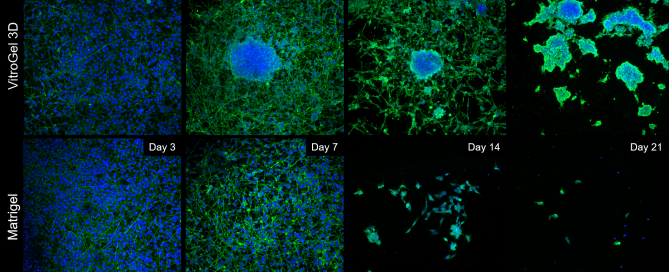
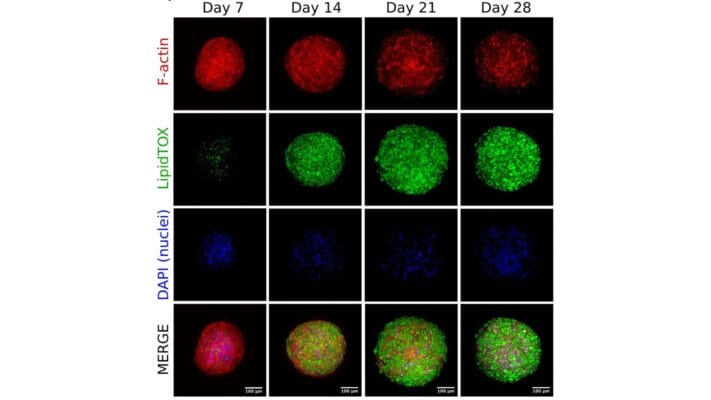
Figure 4. Comparison of long-term neuronal culture seeded onto thick hydrogel mats.
Cells are stained with Beta-III-Tubulin (green) cytoskeleton marker and their nuclei are counter-stained with DAPI (blue). Cells spread out and form neural-like networks as early as day 3 post-differentiation, with comparable efficacy between VitroGel 3D and Matrigel, based on cell survival, culture spreading, and morphological analysis reached between days 7 and 9. On Matrigel mats, cell culture health and viability drop off sharply once day 9 has passed, with most cells detaching and neurites retracting by day 14 and the vast majority of cells gone by day 21. If grown onto VitroGel 3D mats, differentiated B35 neurons have a tendency to self-organize into 3D clusters very early on (Day 7), assuming a mixed 2D/3D cell culture for the first two weeks of the time course. By Day 21, these cells have migrated into self-assembled 3D clusters, embedded into the thick hydrogel matrix, with very few cells between the clusters, but without any significant cell death.
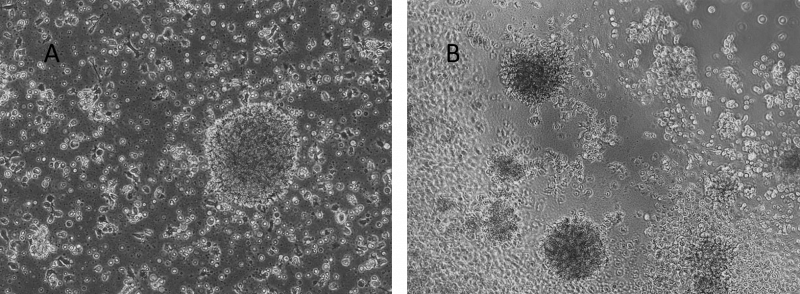
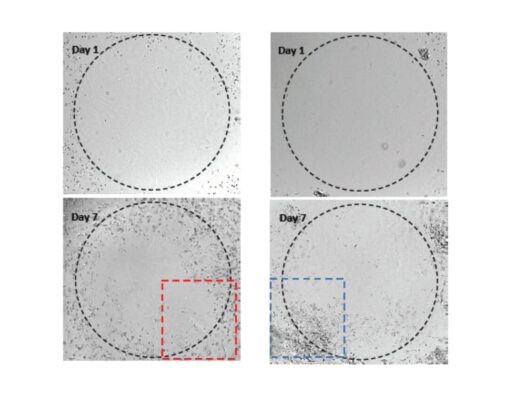
Figure 5. Human Lymphoblastoid Priess cells cultured on top of VitroGel 3D hydrogel.
A. Priess cells grown in suspension (control); B. Priess cells grown on top of VitroGel 3D at day 7. A hydrogel substance can be prepared with different stiffness by adjusting the dilution of VitroGel 3D from 1:1 to 1:3 ratio. Cells seeded on the top of the hydrogel form cell spheroids form on the top of the hydrogel. The hydrogel provides a soft substance for cells to attach and grow.
References/Publications
- Zhou, H., Zhang, Z., Liu, Z., Sa, G., Jiang, M., Zou, Z., Shi, Y., Zheng, L., Yang, X., & Sa, G. (2025). Single‐Cell Analysis Reveals Fibroblast‐Derived Migrasomes as CXCL12 Carriers Promoting Skin Wound Repair. Journal of Extracellular Vesicles, 14(6). https://doi.org/10.1002/jev2.70112
- Sun, P., Qin, W., Xu, H., Yin, H., Yang, L., Zhang, X., Jin, X., Xu, Q., Wu, H., Xiaoling Kuai, Jia, L., Huang, J., & Wang, Y. (2025). SPTSSA facilitates gastric cancer progression with modulating PD-L1 in immunomicroenvironment through Wnt/β-catenin pathway. Cellular Oncology. https://doi.org/10.1007/s13402-025-01072-7
- Chen, Z., Zheng, X., Mu, Z., Lu, W., Zhang, H., & Yan, J. (2025). Intelligent Nanomaterials Design for Osteoarthritis Managements. Small Methods. https://doi.org/10.1002/smtd.202402263
- Chang, Y. W., Trimp, M., Van Der Helm, T., Blanch-Asensio, A., Overeem, A. W., & Chuva De Sousa Lopes, S. M. (2025). Reconstitution of human fetal ovaries reveals niche requirements for primordial germ cell-like cell progression. https://doi.org/10.1101/2025.03.21.644608
- Dariolli, R., Nir, R., Mushlam, T., Souza, G. R., Farmer, S. R., & Batista, M. L. (2025). Optimized scaffold-free human 3D adipose tissue organoid culture for obesity and disease modeling. SLAS Discovery, 31, 100218. https://doi.org/10.1016/j.slasd.2025.100218
- Castañeyra-Ruiz, L., Lee, S., Chan, A. Y., Shah, V., Romero, B., Ledbetter, J., & Muhonen, M. (2022). Polyvinylpyrrolidone-Coated Catheters Decrease Astrocyte Adhesion and Improve Flow/Pressure Performance in an Invitro Model of Hydrocephalus. Children, 10(1), 18. https://doi.org/10.3390/children10010018
- Wei, J., Yao, J., Yang, C., Mao, Y., Zhu, D., Xie, Y., Liu, P., Yan, M., Ren, L., Lin, Y., Zheng, Q., & Li, X. (2022). Heterogeneous matrix stiffness regulates the cancer stem-like cell phenotype in hepatocellular carcinoma. Journal of Translational Medicine, 20(1). https://doi.org/10.1186/s12967-022-03778-w
- Yu, Y., Wu, X., Wang, M., Liu, W., Zhang, L., Zhang, Y., Hu, Z., Zhou, X., Jiang, W., Zou, Q., Cai, F., & Ye, H. (2022). Optogenetic-controlled immunotherapeutic designer cells for post-surgical cancer immunotherapy. Nature Communications, 13(1), 6357. https://doi.org/10.1038/s41467-022-33891-9
- Manferdini, C., et al. (2022). RGD-Functionalized Hydrogel Supports the Chondrogenic Commitment of Adipose Mesenchymal Stromal Cells Gels. https://www.mdpi.com/2310-2861/8/6/382
- Ouyang,L., et al.(2022) Overexpressing HPGDS in adipose-derived mesenchymal stem cells reduces inflammatory state and improves wound healing in type 2 diabetic mice. Stem Cell Research & Therapy, 2022,13:395. https://stemcellres.biomedcentral.com/articles/10.1186/s13287-022-03082-w
- Worden, Austin N.(2022) A novel model to study adipose-derived stem cell differentiation. University of South Carolina ProQuest Dissertations Publishing, 2022, 28967872. https://www.proquest.com/openview/726a089f8f0894146e3f9bf083913e3a/1?pq-origsite=gscholar&cbl=18750&diss=y
- Fen, et al.(2022) Optimization of Three-Dimensional Culture Conditions of HepG2 Cells with Response Surface Methodology Based on the VitroGel System. Biomedical and Environmental Sciences, 2022,(35,8), 688-698. https://www.frontiersin.org/articles/10.3389/fimmu.2022.914381/full
- Yamazaki et al.(2022) Assessment of hypoxia-targeting therapy for intestinal T-cell lymphoma in dogs: preclinical test using murine models. Available at SSRN: https://ssrn.com/abstract=4090297 or http://dx.doi.org/10.2139/ssrn.4090297
- Cui, J., et al. (2022). ATR inhibition sensitizes liposarcoma to doxorubicin by increasing DNA damage. American Journal of Cancer Research. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9077062/
- Fengyuan, M. Y., Shen, L., Fan, D. D., Bai, Y., Li, B., & Lee, J. (2022). YAP9/A20 complex suppresses proinflammatory responses and provides novel anti-inflammatory therapeutic potentials. Frontiers in Immunology. https://www.frontiersin.org/articles/10.3389/fimmu.2022.914381/full
- Sinjushin, A., et al. (2022). Variations in Structure among Androecia and Floral Nectaries in the Inverted Repeat-Lacking Clade (Leguminosae: Papilionoideae) Plants, Special Issue: Floral Secretory Tissue: Nectaries and Osmophores. https://www.mdpi.com/2223-7747/11/5/649
- Worden, A., et al. (2022). Self-Assembling Toroidal Cell Constructs for Tissue Engineering Applications Microscopy and Microanalysis. https://doi.org/10.1017/S1431927622000253
- Chen, Y., et al. (2021). Ultra-sensitive responsive near-infrared fluorescent nitroreductase probe with strong specificity for imaging tumor and detecting the invasiveness of tumor cells Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. https://doi.org/10.1016/j.saa.2021.120634
- Powell K.(2017) Adding depth to cell culture. Science, 356(6333), 96–98. https://doi.org/10.1126/science.356.6333.96
| High Concentration Kit Type | VitroGel 3D + Dilution Solution TYPE 1, VitroGel 3D + Dilution Solution TYPE 2 |
|---|