Research Highlights
VitroGel Hydrogel Used to Bridge the Gap Between In Vivo and In Vitro Pancreatic Islet Beta-cells Studies
Institution:

Xiamen Medical College
Team:
Tianshu Lan, Jingyi Guo, Xiaoming Bai, Zengjiong Huang, Zhimin Wei, Guicheng Du, GuoliangYan, Lebin Weng, Xue Yi
Application:
Development of 3D islet pancreatic beta-cells (β-cells)
Disease Model:
The mouse β islet cell line, β-TC-6
Hydrogels:
VitroGel® RGD, VitroGel® 3D, VitroGel® Cell Recovery Solution
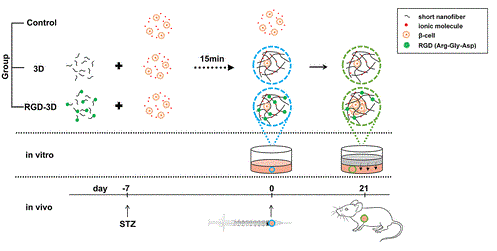
Islet transplantation is currently the only firmly established treatment for diabetes. However, the scarcity of human pancreas donors and the short survival time of islet beta cells (β-cells) after isolation not only limits islet transplantation in vivo, but also in vitro diabetes research. Consequently, many researchers have focused on attempting to build three-dimensional (3D) scaffolds to mimic the in vivo environment post-transplantation islets to enhance their functionality. Although several scaffold materials have been reported as viable candidates, a clinically applicable one that is injectable and can maintain long-term functionality and survival of islet pancreatic beta-cells (β-cells) is far from being established. Many 3D scaffolds cannot be used in both in vitro and in vivo settings, meaning they cannot be used to conduct crossover studies, particularly in tissue engineering.
As a result, Lan and his colleagues evaluated VitroGel hydrogels to support β-cells’ function and viability, both in vitro and in vivo. The viability of the cells was measured by cell proliferation and survival ratio (in vitro) and tissue volume (in vivo). The functions in blood glucose control and insulin secretion were also evaluated. Their results showed that RGD can dramatically increase the survival rate and insulin secretory capacity of β-TC-6 by preventing cell apoptosis. Moreover, our hydrogel is biocompatible, which enables it to maintain β-cell proliferation and vascularization without stimulating inflammation after subcutaneous injection. In the β-cell injection model, the researchers found that direct contact of the cells with RGD in the matrix was especially important, which is also supported by the fact that RGD can interact with integrin in bone marrow cells. These preliminary results indicate that our RGD-modified hydrogel is a potential extracellular matrix for islet transplantation at extrahepatic sites and that they also provide a reference for future tissue engineering studies.
Read the publication:
Related Products: