Application Notes
New hPSCs Expansion Method – Direct from Liquid Nitrogen to 3D Culture Using a Xeno-free Hydrogel – VitroGel® STEM
Culturing and maintaining human pluripotent stem cells (hPSCs) directly from cryopreservation using the xeno-free, hydrogel-based VitroGel® STEM
Application Note
TheWell Bioscience, North Brunswick, NJ 08902
Introduction
Due to the advent of stem cell technologies, there has been an unprecedented rise in the use of human pluripotent stem cells (hPSCs) in biomedical research. Because of its several unique biological properties, such as the ability to self-renew and differentiate into numerous cell types, hPSCs are highly suitable for studying human development, model complex human diseases as well as serve as tools for regenerative medicine. hPSCs also have great potential in drug discovery, screening, and development.
It is important to acknowledge that the effectiveness of hPSCs for biomedical research correlates with the quality of cells, which in turn relates to culturing methods. Conventional methods of maintaining and culturing hPSCs involve the use of 2D matrix-coated culture vessels. Several types of matrices are available commercially today, some of which are of animal origins, while others are derived from transformed cells. A majority of these matrices are expensive, and the process of preparing the matrices and coating them is time-consuming. Because of the smaller surface area of conventional 2D cultures, there is limited to how much the hPSC cultures can be scaled up. Moreover, many of these matrices are temperature sensitive, resulting in instances where the matrix is not coated evenly on surfaces, reducing the quality and uniformity of the hPSC colonies. Due to the fragility of hPSC cells, the viability of hPSCs is negatively impacted upon thawing from cryopreserved vials on 2D culture. Additionally, in order to release hPSC colonies from the surfaces of the culture vessels, harsh methods (enzymatic or non-enzymatic) have to be utilized, which negatively affects cell viability.
In recent years, advancements in stem cell and 3D culture technologies have promoted the use of 3D cell cultures in stem cell and organoid research. For instance, 3D neuro-spheroids can be generated from hPSCs to study complex human diseases such as Alzheimer’s Disease1. Also, the increasing interests in exploring stem cell-based therapies for cellular therapies and regenerative medicine have led to large-scale, 3D-based production of hPSCs2. This simply cannot be achieved through current 2D culturing methods. Therefore, there is a pressing need to devise strategies to adapt current hPSC cultures into 3D systems in order to generate spheroids or organoids with improved efficiency and quality. The ability to culture hPSCs as 3D spheroids is the first step to achieving this milestone.
Several studies have already been published highlighting the utility of hydrogels to generate 3D hPSC cultures3–5. However, at present, there is still a lack of 3D hydrogel-based methods to culture and maintain hPSCs at a large scale, especially directly from cryopreserved vials. This would become an obstacle to standardizing downstream 3D applications such as organoid formation and large-scale stem-cell-derived production for therapeutic purposes.
Herein, we show that hPSCs can be easily cultured and maintained in the xeno-free, hydrogel-based VitroGel® STEM (Cat No. VHM02) as 3D spheroids in static conditions directly from Liquid Nitrogen. 3D spheroids can be passaged, and the sizes of these spheroids can also be readily controlled. VitroGel STEM is easy to use, and it ensures high quality of spheroids that are easily harvested of subcultured. The utility of VitroGel STEM is highly cost-effective since it eliminates the need for matrix coating or microcarriers and expensive laboratory shakers and stirrers. After culturing hPSCs in VitroGel STEM, hPSCs retain high levels of pluripotency markers and remain highly viable. Figure 1 shows a summary of how VitroGel STEM can be used to culture and maintain hPSCs as 3D spheroids either through a 3/4- day culture cycle or a 7-day culture cycle just by simply mixing cells with the hydrogel solution (Figure 2). Media changes are less frequent, allowing ease for hPSC culture and cell harvesting.
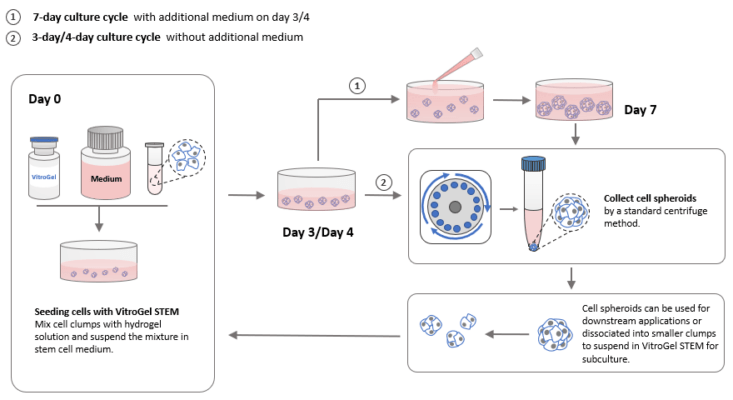
Figure 1. Schematic diagram summarizing how 3D hPSC spheroids can be cultured in suspension using VitroGel STEM.
Materials and Methods
Forming 3D Spheroid Cultures from Frozen Vials of hPSCs
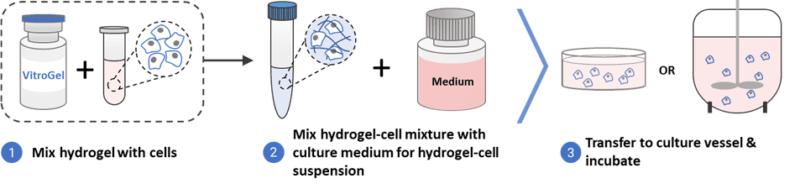
Figure 2. 3D hPSC spheroids can be generated by mixing hPSCs with VitroGel STEM.
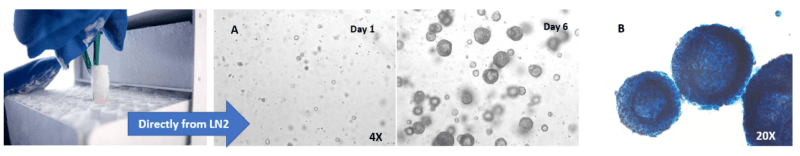
3D hPSC spheroids can be made from cells directly from frozen liquid nitrogen stocks (Figure 3). VitroGel STEM is able to support the recovery and expansion of thawed hPSCs. Conventional methods require the culturing of hPSCs on a matrix for multiple passages before hPSC cell lines recover and stabilize. During the thawing process, preserving the viability of thawed hPSCs is typically challenging. However, VitroGel STEM eliminates that difficulty by allowing 3D spheroids to be generated from cells taken out from liquid nitrogen. Shaking or rocking is not needed for 3D spheroid formation.
Generating 3D spheroids from 2D cell colonies or frozen cells can be done by:
- Bring VitroGel STEM to room temperature or warm up to 37°C.
- Resuspend thawed cells in the appropriate media before centrifuging to obtain cell pellet and remove cell freezing solution.
- Prepare cell suspension in stem cell media with 10μm/mL ROCK Inhibitor Y-27632 *.
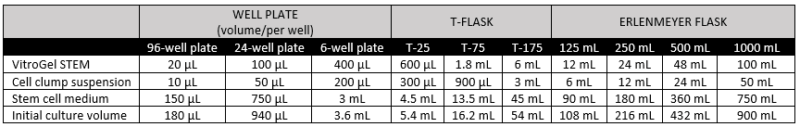
- Gently mix VitroGel STEM with cell suspension at 2:1 (v/v) ratio. (Check Table 1 or Table 2 of the recommended volume of different culture vessels for different culture cycles)
- Add stem cell medium with 10 μm/mL Y-27632 to the cell‐hydrogel mixture at a 5:1 (v/v) ratio. This ratio is critical in ensuring hPSC spheroids are well-formed. Carefully pipette up and down to mix the medium and mixture homogeneously**.
- Add the desired volume of the mixture to the culture vessel and incubate at 37°C with 5% CO2 (See Table 1 and 2).
- Add additional medium for 7‐day culture cycle: On day 3 or day 4, add the desired volume of stem cell medium (without Y‐27632) directly to the culture vessel (See Table 2).
* The recommended cell density is 0.2‐5 X 106 cells/mL for the final cell seeding density in the hydrogel suspension culture around 0.1‐5 X 105 cells/mL. The cell seeding density should be optimized for individual cell types and will also depend on the desired final sizes of the stem cell spheroid.
** The mixing ratios of stem cell medium and cell‐hydrogel mixture can be adjusted between 1:1 to 20:1 v/v ratio, depending on the desired viscosity of the final mixture and culture conditions. If the mixing ratio is higher than 5:1 v/v, an orbital shaker or spin flask may be required and set at a speed of 10‐40 rpm to maintain the cell suspension.
3D hPSC spheroids can also be made from hPSCs cultured in conventional 2D monolayers. If monolayer hPSC cultures are used, dissociate cells using a suitable dissociation reagent (as per manufacturer’s recommendations) and then continue from steps 3 onwards.
Figure 3. 3D hPSC spheroids can be established directly from cells stored in liquid nitrogen (LN2).
A. hPSC spheroids formed directly from cells stored in liquid nitrogen grow continuously from day 1 to 6. B. Alkaline Phosphatase (AP) staining shows that these spheroids are healthy and that they retain their pluripotency indicated by high levels of AP expression.
Table 1. Recommended volume for 7-day culture cycle
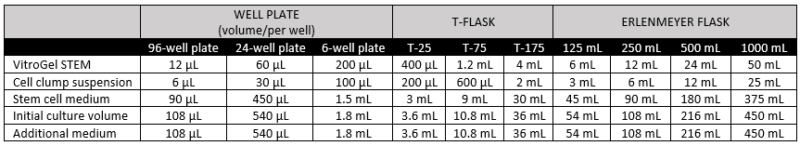
Table 2. Recommended volume for 3-day or 4-day culture cycle
Harvesting hPSC spheroids from VitroGel STEM
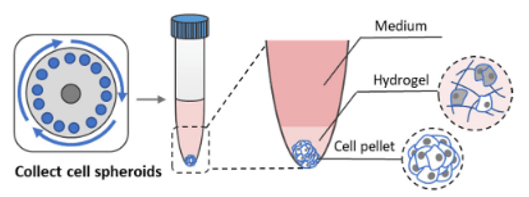
Figure 4. Diagram illustrating the hydrogel layer and cell pellet in the conical tube after centrifugation
hPSC spheroids formed directly from cells stored in liquid nitrogen grow continuously from day 1 to 6. B. Alkaline Phosphatase (AP) staining shows that these spheroids are healthy and that they retain their pluripotency indicated by high levels of AP expression.
- Transfer hPSC spheroids from the culture to a conical tube by using a serological pipette.
- Centrifuge the tube for 3 minutes at 100 x g.
- Carefully remove supernatant and hydrogel layer and collect cell pellet.
If cells in the hydrogel layer are needed (Figure 4), skip step 3 and proceed to step 4.
- Carefully remove supernatant
- Add VitroGel® Cell Recovery Solution (SKU: MS03‐100) to the tube (warm the solution at 37 °C before use). Keep the volumes of cell recovery solution and cell pellet/hydrogel mixture at 5:1 (v/v) ratio.
- Gently mix with a serological pipette and incubate at 37 °C for 3‐5 minutes.
- Centrifuge the tube for 3 minutes at 100 x g to collect the additional cell pellet.
Passaging 3D hPSC Spheroids
3D hPSC spheroids cultured in VitroGel STEM can be easily passaged using cell dissociation solution. This can be done by:
- Transfer hPSC spheroids from the culture to a conical tube by using a serological pipette.
- Centrifuge the tube for 3 minutes at 100 x g.
- Carefully remove supernatant and hydrogel layer and collect cell pellet (If cells in the hydrogel are needed, please refer to ‘Harvesting hPSC spheroids from VitroGel® STEM’)
- Add cell dissociation solution (as per manufacturer’s recommendations) to the tube and resuspend cells. Incubate the mixture at 37 °C for 3‐5 min.
- Centrifuge the tube for 3 minutes at 100 x g to collect the cell pellet.
- Add the desired volume of stem cell medium (with 10 μm/mL Y‐27632) to resuspend the cells. Use a pipettor with a 1 mL pipette tip, carefully pipette up and down to break up the clumps to 30‐70 μm in size (Figure 5).
- The cells are ready to be mixed with VitroGel STEM (please refer to ‘Forming 3D spheroid cultures from 2D monolayer cells or frozen vials’)
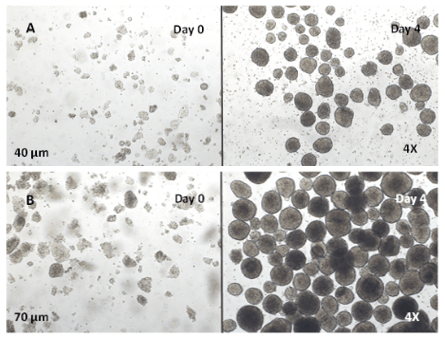
Figure 5. Passaging of hPSC spheroids of different sizes
A. hPSC spheroids were broken down into 40μm sized clumps. B. hPSC spheroids were broken down into 70 μm sized clumps. After 4 days in culture, the cell clumps of either size grew into spheroids with different sizes depending on the size of the initial cell clumps.
Re-establishing 2D hPSC Colonies
If desired, stem cell spheroids can also be used to re-establish 2D culture by adding on 2D matrix coated tissue vessels (Figure 6). This can be done by first harvesting cells from the hPSC spheroids as per ‘Harvesting hPSC spheroids from VitroGel® STEM.’ After which, the cells can be resuspended in stem cell medium (with 10 μm/mL Y‐27632) and seeded on a matrix-coated surface (as per manufacturer’s recommendations).

Figure 6. Re-establishing 2D hPSC colonies from spheroids
A. hPSC spheroids Cells from those hPSC spheroids seed and attach well onto a matrix-coated surface. Those cells have grown and expanded into large 2D hPSC colonies.
Viability Analysis
The viability of 3D hPSC spheroids over time is measured using the Cyto3D™ Live-Dead Assay Kit (Cat. No. BM01). The assay can be performed by:
- The Cyto3D Live-Dead Assay Kit was brought to room temperature.
- Add 2 µL of Cyto3D reagent to every 100 µL total volume in a well.
- Incubate the cells at 37°C for 5-10 minutes. The cells are ready for cell viability detection.
- The cells were imaged on a fluorescence microscope: live cells were observed using GFP filter while dead cells were observed using Texas red filter.
Results and Discussion
VitroGel STEM supports hPSC growth and expansion
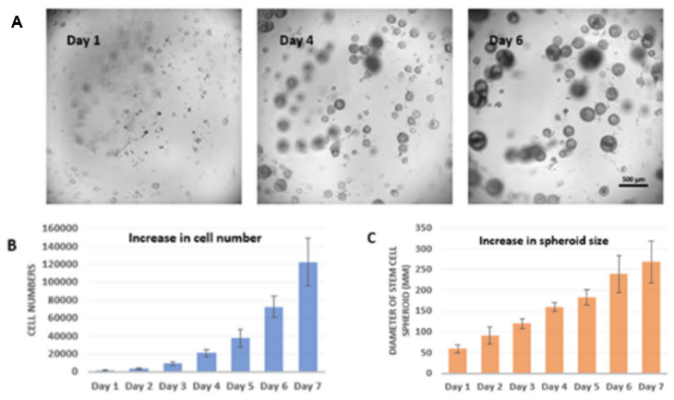
Figure 7. 3D hPSC spheroids cultured for 7 days
A. Start the suspension culture by using healthy and high-quality cells from 2D matrix coating culture. The cell spheroids formed and kept growing from day 1 to day 6. The cell spheroid shows characters of shallow craters or pockmarks, which indicate the high expression of hPSC markers and good expansion of cells at the healthy and high-quality states. B. The chart shows the growth of the cell number from day 1 to day 7 after being seeded for 3D static suspension culture (The cell proliferation assay was tested by CCK8 assay). C. The increase of cell sizes of hPSC spheroids from around 60µm at day 1 to over 250 µm at day 7.
Effective Control of Spheroid Cells
hPSC spheroids of different sizes are required for different differentiation protocols or downstream applications. VitroGel STEM supports the growth of hPSC spheroids of different sizes. The flexibility of VitroGel STEM allows for control over spheroid size, simply by varying cell densities used to generate the spheroids. Figure 8 shows that after 3 days of culture, spheroids of varying sizes expand in size. In addition, the higher the cell density used to generate the spheroid, the large the spheroid size.
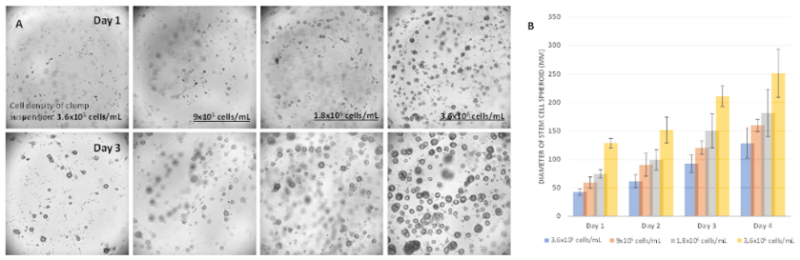
Figure 8. hPSC spheroids of different sizes generated from different cell densities
A. The morphology of the hPSC growth from day 1 to day 3 at initial densities of cell clump suspension at 3.6×105 cells/mL, 9×105 cells/mL, 1.8×106 cells/mL, and 3.6×106 cells/mL, respectively. The initial sizes of the cell spheroids are different according to different cell seeding densities. B. The increasing of cell sizes of hPSC spheroid with different initial cell seeding densities.
VitroGel STEM Maintains Pluripotency of hPSCs
One of the key concerns when it comes to culturing hPSCs is unwanted spontaneous differentiation resulting in the loss of pluripotency. This will negatively impact downstream applications, including gene editing, tissue organoid development, cell reprogramming, biochemical assays, and DNA/RNA sequencing due to the reduced purity of differentiated cell populations. This has especially significant implications in stem cell-based therapies where the purity of differentiated cells is crucial in restoring tissue functions. Immunofluorescence staining on the 3D hPSC spheroids cultured in VitroGel STEM for 7 days shows that spontaneous differentiation was not induced. The 3D hPSC spheroids retain high expression levels of pluripotency markers SSEA, OCT4, SOX2, and TRA-1-60 (Figure 9).
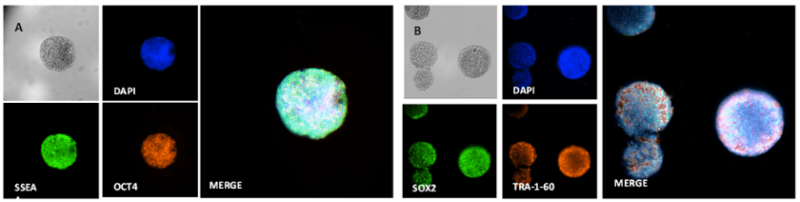
Figure 9. hPSC spheroids of different sizes generated from different cell densities
A. Immunofluorescence staining shows that 3D hPSC spheroids cultured for 7 days express SSEA and OCT4. B. Immunofluorescence staining shows that the 3D hPSC spheroids express SOX2 and TRA-1-60.
VitroGel STEM Promotes Cell Viability of hPSCs
hPSCs possess unique characteristics that make them highly attractive for biomedical research –they are highly proliferative and capable of self-renewal. Therefore, it is important that the method by which hPSCs are cultured and maintained does not stifle cell viability. Using the Cyto3D™ Live-Dead Assay Kit (Cat No. BM01), which identifies live and dead cells due to preferential staining, it was shown that the 3D hPSC spheroids cultured in VitroGel STEM are composed of live cells (Figure 10). Minimal dead cells (stained by propidium iodide) were observed. VitroGel STEM is highly compatible with different types of stem cell media to support hPSC growth and expansion.
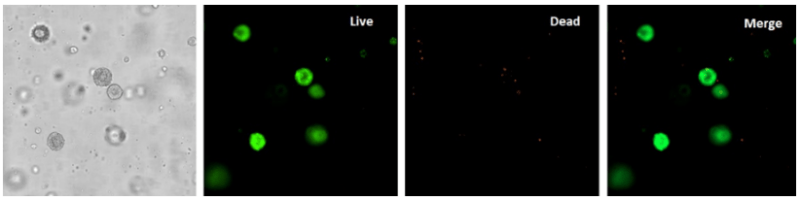
Figure 10. 3D hPSC spheroids are composed of living cells
Live/Dead assay performed, using the Cyto3D Live-Dead Assay Kit, on 3D hPSC spheroids show that the spheroids are composed of live cells stained by the Acridine orange dye. Minimal dead cells, stained by propidium iodide, were observed.
In summary, this study illustrates the suitability of xeno-free, hydrogel-based matrix VitroGel® STEM to routinely culture and maintain hPSCs in 3D spheroids. hPSCs retain their high proliferative properties, allowing the 3D hPSC spheroids to expand and grow in size. The flexibility of VitroGel STEM allows spheroids of different sizes to be generated. Lastly, immunostaining analysis confirms that the 3D hPSC spheroids maintain high levels of pluripotency and cell viability. This provides confidence that VitroGel STEM facilitates hPSC growth and ensures consistent and high-quality cells are produced for stem cell and biomedical research.
Reference
- Lee, H.-K. et al. Three Dimensional Human Neuro-Spheroid Model of Alzheimer’s Disease Based on Differentiated Induced Pluripotent Stem Cells. PLOS ONE 11, e0163072 (2016).
- Kropp, C., Massai, D. & Zweigerdt, R. Progress and challenges in large-scale expansion of human pluripotent stem cells. Process Biochemistry 59, 244–254 (2017).
- Lei, Y. & Schaffer, D. V. A fully defined and scalable 3D culture system for human pluripotent stem cell expansion and differentiation. PNAS 110, E5039–E5048 (2013).
- Le, M. N. T. & Hasegawa, K. Expansion Culture of Human Pluripotent Stem Cells and Production of Cardiomyocytes. Bioengineering 6, 48 (2019).
- Shao, Y., Sang, J. & Fu, J. On human pluripotent stem cell control: The rise of 3D bioengineering and mechanobiology. Biomaterials 52, 26–43 (2015).