Webinars
Development of In Vitro Intestinal Model With Macrovascular Endothelium: Compare Animal-based and Xeno-free ECM Platforms
On-Demand WeBINAR
SPEAKER

Scarlett Zeiringer
University of Graz
Institute of Pharmaceutical Sciences

WATCH THE WEBINAR
Scarlett Zeiringer is a PhD student at the University of Graz, Institute of Pharmaceutical Sciences, Austria.
She works in Prof. Eva Roblegg’s lab at the Institute of Pharmaceutical Technology and Biopharmacy, where she is working on in vitro cell models to study the fate of drug delivery systems. In particular, she aims to develop advanced xeno-free, cell-based systems that mimic the intricate physiology of the small intestine. By incorporating complex physiological features, these models provide invaluable insights into drug delivery behavior, leading to more efficient and ethical drug development practices.
Webinar Summary:
Development of In Vitro Intestinal Model with Macrovascular Endothelium: Compare Animal-based and Xeno-free ECM Platforms
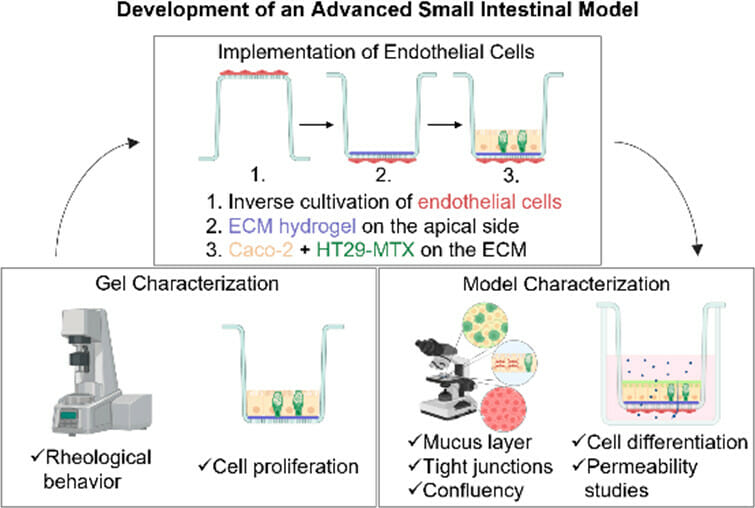 In vitro intestinal models serve as vital tools for studying biological processes, drug absorption and cytotoxicity while reducing the need for animal testing. Typically, these models consist of enterocytes and mucus-producing cells that are cultured for three weeks and closely mimic the human epithelium. Key components also include the extracellular matrix (ECM) for structural support and promotion of cellular processes, as well as a robust vascular network consisting of endothelial cells for transport and immune cell coordination. However, the use of animal-derived hydrogels in these models can lead to misleading results and the endothelium is rarely considered at all. To address this, we investigated synthetic, customizable hydrogels such as VitroGel® and Peptigel® as alternatives to animal-derived ECMs such as Matrigel®, performing rigorous rheological characterization and investigating their effect on cell proliferation. The results led to the development of an advanced in vitro intestinal model that favors the xeno-free VitroGel® as the ECM and includes the endothelium, providing a more reliable and animal-free approach.
In vitro intestinal models serve as vital tools for studying biological processes, drug absorption and cytotoxicity while reducing the need for animal testing. Typically, these models consist of enterocytes and mucus-producing cells that are cultured for three weeks and closely mimic the human epithelium. Key components also include the extracellular matrix (ECM) for structural support and promotion of cellular processes, as well as a robust vascular network consisting of endothelial cells for transport and immune cell coordination. However, the use of animal-derived hydrogels in these models can lead to misleading results and the endothelium is rarely considered at all. To address this, we investigated synthetic, customizable hydrogels such as VitroGel® and Peptigel® as alternatives to animal-derived ECMs such as Matrigel®, performing rigorous rheological characterization and investigating their effect on cell proliferation. The results led to the development of an advanced in vitro intestinal model that favors the xeno-free VitroGel® as the ECM and includes the endothelium, providing a more reliable and animal-free approach.
Join Scarlett Zeiringer from the University of Graz, Institute of Pharmaceutical Sciences, to present their findings.
Key Takeaways:
- Workflow of the development of a xeno-free in vitro intestinal model that considers the macrovascular endothelium.
- Understanding the advantages and the opportunities of using VitroGel® compared to others.
- Providing a platform to study drug transport/efflux across the intestinal barrier.