Webinars
Robust and Reproducible Expansion of Human Mesenchymal Stem Cells using RocketCell™ hMSC Xeno-Free Complete Medium
Learn how a standardized xeno-free platform enables a robust, multipotent MSC expansion suitable for translational application.
Human mesenchymal stem cells (hMSCs) have evolved from an early cytological discovery into a cornerstone of regenerative medicine, with over 70,000 publications supporting their therapeutic potential. First described as colony-forming unit fibroblasts by Alexander Friedenstein and later defined as “mesenchymal stem cells” by Arnold Caplan in 1991, hMSCs are known for their ability to differentiate into bone, cartilage, and fat. More recently, their role has expanded beyond differentiation, with recognition as “Medicinal Signaling Cells” due to their powerful immunomodulatory effects, trophic secretome, and exosome production. Their unique immunoprivileged properties and ability to modulate immune responses and promote tissue repair have enabled their application in treating a wide range of conditions, including graft-versus-host disease, autoimmune disorders, osteoarthritis, spinal cord injury, and other degenerative and inflammatory diseases. These advances, along with the discovery of related cell populations such as MAPC and MUSE cells, highlight the growing need for robust, reproducible stem cell culture systems to support research and clinical translation.
However, a significant barrier to widespread clinical translation remains the historical reliance on fetal bovine serum (FBS) in expansion protocols. The lack of standardized, xeno-free (animal-product-free) culture media presents substantial regulatory and safety challenges. The use of xenogenic supplements introduces risks of prion transmission, viral contamination, and eliciting host immune responses against bovine proteins, which complicate FDA approval. Until chemically defined, xeno-free media are universally adopted to ensure batch-to-batch consistency and patient safety, the transition from “bench to bedside” will remain a complex and costly regulatory hurdle for the global regenerative medicine industry.
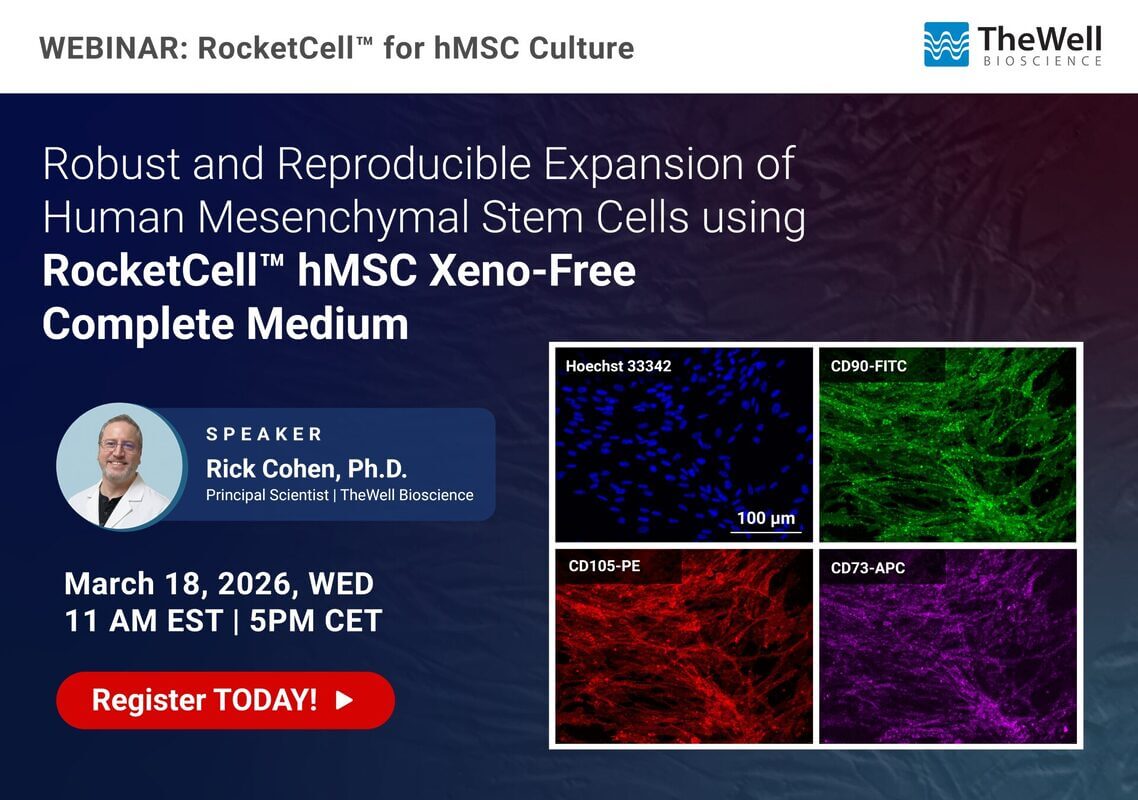
In this webinar, we will present data demonstrating that RocketCell™ hMSC Xeno-Free Complete Medium supports robust MSC expansion with minimal adaptation from traditional serum-containing conditions, including recovery and growth of long-term cryopreserved bone marrow–derived hMSCs. The medium enables efficient low-density expansion, with or without extracellular matrix support, while preserving MSC identity and multipotency, as confirmed by expression of canonical markers and by standard tri-lineage differentiation into adipogenic, chondrogenic, and osteogenic lineages. These results highlight a reliable, xeno-free solution for standardized MSC culture and translational applications.
KEY TAKEAWAYS
- Xeno-Free hMSC culture provides a more physiologically relevant foundation for disease modeling, secretome studies, exosome production, and translational research than traditional systems that contain animal products.
- The RocketCell™ hMSC Xeno-Free Complete Medium delivers a standardized, all-in-one solution that supports rapid expansion while maintaining multi-potency, long-term viability, and reproducibility.
- Together with our VitroPrime™ 3D Culture Vessels and CytoGrow™ Fibronectin, the RocketCell™ hMSC Xeno-Free Complete Medium enables researchers to transition from other animal-containing media with minimal effort.
PRESENTER

Rick Cohen, Ph.D.
TheWell Bioscience
Principal Scientist
Dr. Rick Cohen is a stem cell biologist with more than two decades of experience in stem cell research, iPSC technologies, and advanced cell culture systems. He received his doctorate in Pharmacology and Therapeutics from McGill University and completed postdoctoral training in neurogenetics at the NIH (NINDS).
Over the course of his career, Dr. Cohen has established standardized stem cell culture protocols, developed viral and non-integrating methods for iPSC generation, and engineered functional differentiation systems across multiple lineages. He has trained more than 100 scientists in human stem cell techniques and has contributed extensively to stem cell biology, including work on iPSC expansion, neuronal differentiation, and scalable culture systems.
His expertise spans both foundational stem cell biology and translational applications, with a strong focus on developing reproducible and physiologically relevant culture platforms.